Propagation of Corylus
(Hazelnut, Filbert, Cobnut)



By Aimee Stockman
Due to my overwhelming interest in the tasty Corylus nut, you are invited on a journey to learn about the production and propagation of Hazelnut and the trees that produce them.
Introduction
Hazelnuts come from the family Corihave. There are about 18 different species of Corylus. The term 'hazel' comes from the old English word for hood. Some believe that the term filbert, another common name, came from the nut’s long husk being referred to as a “full beard” in Old English. Dictionaries refer to the cobnut as a game that used to be played by children with the nuts.
Corylus avellana is produced in Turkey, Italy, Spain, France, and the United States. Turkey dominates this market in the world, with an astounding 70% compared to other countries. Italy is second; the United States, third; China, fourth; Spain, which had the third largest crop five to ten years ago, has reduced its acreage and is now fifth. The number one producer of hazelnuts in the U.S. is Oregon.
In the United States hazelnuts are marketed both unshelled and shelled in raw, roasted, or salted forms. They are used in the confectionery industry and in a wide range of food products.
Methods of Propagation
I have found that the hazelnut tree is clearly out of the ordinary in a horticultural sense. It has a unique and wonderful side to it. The way in which it achieves pollination in the winter and completion of nut set in the spring, is truly interesting. The female flowers on the tree are flowers that have stigmas on the ends, while the male flowers are the catkins. The catkins are able to shed pollen.
Pollinators should be available in the nurseries as peak periods of male and female flowering may not coincide for any one variety. This will ensure cross-pollination.
The following are the main types of propagation and a little information about each:
Cuttings
The use of leafy cuttings with bottom heat in a coarse rooting medium and indole butyric acid (IBA) hormone treatment has had limited success. High humidity ventilated fog has been found to increase the rooting percentage of hazelnut leafy cuttings; however, they are also more susceptible to basal necrosis with this method.
Seeds
Hazelnuts do not reproduce true to type from seed. Collect nuts from the trees rather than from the ground when they begin to turn brown. The nuts will fall when they turn fully brown however they do not usually last long as wildlife will clear the floor of fallen nuts. Use the flotation test to see if the seed is viable.
Hazelnut seeds have a hard seed coat, internal dormancy and irregular germination with increasing length of storage. To overcome this dormancy, the following procedure is followed for C. avellana in Oregon. Half-brown nuts are harvested in August and are refrigerated. In late November, the nuts are soaked for two to four days in water then stratified in moist vermiculite at 4oC for three to five months. After three months the seeds are warmed for 5 days and those with visible root tips are planted in flats in the greenhouse. Ungerminated seeds receive further stratification. Seedlings are transplanted after they reach 25 cm.
Another way to overcome this dormancy is to
soak the seeds in gibberellic acid (GA). The shells would have to be removed
and the kernels
would be soaked in about 25 to 50 ppm GA3 for 16 to 24 hours. Seeds can
then be germinated by planting directly into flats in the greenhouse or
by
placing
on moist filter paper in germinating boxes. The latter method is said
to be preferred so that the seeds that fail to germinate initially can be
stimulated by a second GA treatment. Some seeds, such as
C.
avellana
cv. Gem, or forma heterophylla and pendula, tend to decay in stratification
and must be treated with GA. Freshly harvested seeds treated with GA
have the highest germination rate, but germination of stored, non-stratified
seed is enhanced.
Grafting
Turkish tree hazel is a good rootstock for all of the hazels, mainly because it is non suckering. Its deep taproot results in increased drought tolerance. However, seeds of this species are difficult to germinate and, because of the taproot, seedlings are difficult to transplant. Only vigorous varieties of hazelnut should be grafted onto the Turkish tree hazel as the rootstock tends to outgrow all but the most vigorous varieties.
Hazelnuts begin to bear at approximately three years old and at six years yields should approach 2–2.5 kg/height Grafting can be difficult with Corylus.
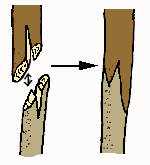 Whip and
tongue graft
Whip and
tongue graft
A whip and tongue graft may be performed in mid-winter. Care is taken to insure that the rootstock and scion are completely dormant prior to grafting. If the rootstock has sap moving upwards in the phloem, a successful callus bridge will not form between the stock and scion.
The surface areas
of the stock and scion must be cut cleanly. It is essential to make sure
that the blade is not dull when cutting either the rootstock or the scion.
A budding rubber or budding tape, is wrapped securely but not tightly
at the union. The graft is placed
in a warm greenhouse for several weeks while the graft union becomes
secure. Later the plant is slowly introduced to harsher conditions and finally
planted
in its natural environment.
Tip Layering
The main method for propagation is by the use of stool beds to produce trees on their own roots. Suckers are girdled and can be treated with a rooting hormone to encourage root initiation. In other cases, more severe disruption is necessary. Inhibited callus formation has prevented grafting in the past but the use of the hot callusing tube has increased grafting success up to 90%.
In simple layering of hazelnut, simply reversing the polarity of the stem is usually sufficient to stimulate rooting. In simple layering, shoots are placed in contact with the ground and secured there so that the shoot tip remains above ground. Commercially, one year-old shoots are layered just prior to bud break, as in hazelnut. By the following autumn, most shoots should be rooted. The suckers arising from the base of vigorous young trees that are four to eight years old, are layered in early spring. The basal portion of suckers are scarred or a hog-nose ring is applied encircling the stem of the young shoots. A 0.5 m tall tarpaper collar is placed at the base of the suckering tree. Sawdust is added to the brim of the tarpaper. Roots form on the suckers during the summer. The tarpaper and sawdust are removed in late August and the rooted layers can be removed from the tree. Sometimes less-well rooted suckers are placed in a nursery bed for about a year for further root development.
Micropropagation
Hazelnut, corylus, is difficult to micropropagate. A system of tissue culture, called double-phase culture, has greatly improved shoot growth of this clone. An advantage to micropropagation is that all micropropagated trees have clonal rootstock. This ensures uniformity and reduced management needs. Plus the micropropagation method cuts years off of the time is takes to grow a traditional layering bed. For the hazelnut hill nursery, 2003 was the first year they offered micropropagated trees for sale. More research is being done every year to improve and continue is this propagation method, due to the many advantages.
Interesting Tidbit
The Northern Nut Growers Association's Willard G. Bixby, former president, discusses the possibilities of starting a nut arboretum. This arboretum could house the best varieties and become a site for further research in hazelnuts for years to come. This would also allow for better control over propagationof the hazelnuts and a controlled environment for crosses between parental plants.
References
http://oregonhazelnuts.org/ For more information from the oregon hazelnut industry
Northwest Hazelnut For recipes, types of nuts and locations to order click on
Nut Arboretum More on the idea for a by Willard G. Bixby
Food and Agricultural Org of the United Nations. For statistics on crop production
GRIN Genetic Resources Information Network.
Hazelnut breeding and propagation in Romania
NSW agriculture on Hazelnut production
Thompson, M. M., H. B Lagerstedt, and S. A. Mehlenbacher. 1996. Hazelnuts. pp. 125-184, Chapter 3. In: Fruit Breeding, Vol. III: Nuts
Hartman, H. and D. Kester. 1975. Methods and Rootstocks for the Important Fruit and Nut Species: Filbert. p. 554. In: Plant Propagation. Third Edition. Prentice- Hall, Inc. Englewood Cliffs, New Jersey.