
Cell Growth, Cancer and Oncogenes
Cancer Genes in the Human Genome
The Cell Cycle
The field of developmental genetics investigates the genetic basis of the changes in form that an organism passes through during its life cycle. One cellular process that is common throughout these changes in form is cell division. The two cell division events that need to be controlled are the entry into the S-phase when DNA is replicated, and the entry into the M-phase when mitosis occurs. In this regard, two timing events need to be monitored by the cell. These are:
- when to initiate replication (S-phase entry)
- when to begin chromosomal condensations (M-phase entry)
Related to these events are four factors that appear to control the entry into the M-phase.
- The accumulation of a specific cellular mass is a factor for somatic cells. This is called the mass factor.
- Some cells need to obtain a specific growth rate for mitosis to begin. This is called the growth rate factor.
- The time between successive M-phases appears to be controlled by timer or oscillator genes. This is the time factor and appears to be a factor in embryo cells.
- The entry into the M-phase also requires completion of the S-phase. This insures that daughter cells receive complete DNA complements and is called the completion of chromosomal replication factor.
For the cell to coordinate these different events, it must be able to monitor the cell cycle. An important biological question that needs to be resolved is how does the cell know where it is in the cell cycle. As you would expect, genetics and biochemical characterization have provided an extensive, but incomplete description of the process.
Cell cycle research has primarily been performed on mutant strains of the fission yeast (Schizosaccharomyces pombe) and the budding yeast (Saccharomyces cerevisae) that have genetic lesions in some phase of the cell cycle. The cell division cycle (cdc) mutant strains have been quite useful in elucidating important steps. The cell cycle in yeast has two points where it is committed to proceed to the next stage in the cycle. The first point called start occurs near the end of the G1, and the cell becomes committed to DNA synthesis in the S phase of the cycle. The second commitment point is at the beginning of the M phase when the cell becomes committed to chromosomal condensation and the subsequent mitotic steps.
The following diagrams illustrate the genetic and biochemical information known about the entry into the M-phase of the cell cycle. As you can see from the biochemical diagram a protein complex is formed at the two committal points. Each complex consists of a protein called cyclin, and a protein kinase called p34. The existence of such a complex was described biochemically when a factor called maturation promoting factor (MPF) was isolated that could initiate mitosis in certain mutant yeast strains whose cell cycle was arrested at this stage. It was the coupling of this type of biochemical research with genetics that defined and elucidated many of the steps in the cycle.
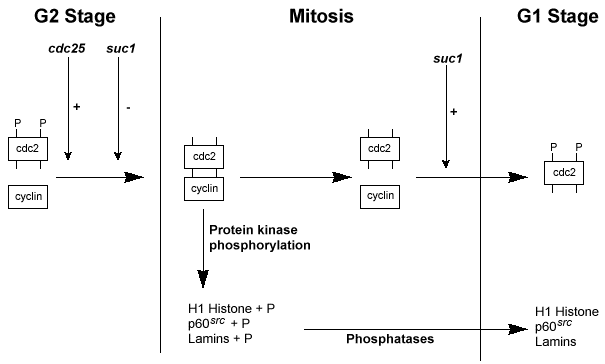
To demonstrate the molecular events associated with the cell cycle in more detail, a discussion that links the genetic research with the biochemical research will be presented. In particular, the discussion will concentrate only on the entry into the M-phase. The following table presents the mutants, the product of these genes and their role in M-phase entry.
| Gene | Protein | Function |
|---|---|---|
| cdc13 | Cyclin | a 45,000-47,000 dalton protein that complexes with the protein kinase p34cdc2 to form the MPF; its sequence is 30% conserved over a 200 amino acid stretch in a wide range of species; M-phase entry can be stimulated by adding this protein from clams to frog cells; its degradation appears to be associated with the inactivation of p34cdc2 |
| cdc2 | p34cdc2 | a serine-threonine protein kinase of 34,000 daltons that complexes with cyclin to form the MPF; the inactive form of the protein is phosphorylated at threonine (T) and tyrosine (Y) residues; the phosphorylation appears to performed by p60src in humans; the active form of the protein is dephosphorylated and it functions by phosphorylating a number of proteins; this phosophorylation activity is coupled to the entry into the M-phase; the protein must be associated with a normal cyclin protein for the M-phase to be completed normally; association with deletion mutants of cyclin halts the M-phase before it is completed |
| cdc25 | p80cdc25 | a protein of 80,000 daltons that assists with the dephosphorylation of p34cdc2 by either inhibiting its phosphorylation or promoting its dephosphorylation; its concentration increases as the cell approaches the M-phase suggesting the accumulation of this protein to a specific concentration is required to activate p34cdc2 ; its increase in concentration appears to be coupled with the completion of the S-phase |
| suc1 | p13suc1 | a protein of 13,000 daltons which may be involved in the inactivation of p34cdc2 late in mitosis by inhibiting its kinase activity or promoting its phosphorylation |
It is clear from the genetic and biochemical studies that the appearance of an active MPF occurs at the M-phase committal point. The following cellular events have been associated with the onset of the protein kinase activity of the cdc2 product.
- chromosomal condensation
- cytoskeletal reorganization
- nuclear envelope breakdown
- cell shape changes
Each of these events is clearly required for cell division to occur. Furthermore, the substrates of the p34cdc2 protein kinase are proteins involved in the maintenance of the cell in the G2-phase. The phosphorylation of these proteins may change their functions and permit the cell to enter the M-phase. The key substrates of p34cdc2 protein kinase are:
- Histone H1 - the phosphorylation of this protein may be important for chromosomal condensation to occur
- Centrosomal protein - these proteins are associated with centrioles, the organizing center of the cell for microtubules associated with the cytoskeleton
- Lamin - this is a protein associated with the nuclear envelope
- p60src - phosphorylation of the mitotic-specific sites of this protein may influence the cytoskeleton and lead to changes in the cell shape
- other DNA binding proteins that need to be released for chromosomal condensation to occur
The studies on yeast and other organisms has lead to the conclusion that a universal control mechanism regulating entry into the M-phase is common to all eukaryotic cells. The key features of the process are as follows.
- The protein kinase activity of p34cdc2 is central to the model. This protein is thought to phosphorylate key proteins that lead to the major events in the M-phase. High levels of this protein maintain the cell in the M-phase, and its inactivation is required for exit from the phase.
- The second key protein is cyclin that complexes with p34cdc2 to form the MPF. Cyclin is required for p34cdc2 activation. Cyclin degradation is required for the cell to exit the M-phase and probably the inactivation of p34cdc2.
- The activation of p34cdc2 is associated with the dephosphorylation of the phosphorylated tyrosine and threonine residues of the protein. Its kinase activity appears to be associated with the tyrosine residue, so dephosphorylating this site appears essential.
- Timing of the M-phase entry is associated with two other protein kinases and the accumulation of p80cdc25. This timing event is associated with the dephosphorylation of p34cdc2.
- p13suc1 interacts with p34cdc2 and may be involved in its rephosphorylation at the end of the M-phase.