The lac Operon - an inducible system
The trp Operon - a repressible system
The trp Operon - a repressible system
The trp operon of E. coli controls the biosynthesis of tryptophan in the cell from the initial precursor chorismic acid. This operon contains genes for the production of five proteins which are used to produce three enzymes. The products of the E and D genes form a multimeric protein comprised of two copies of each protein to produce the enzyme anthranilate synthetase. This enzyme catalyzes the first two reactions in the tryptophan pathway. The next enzyme, which is responsible for catalyzing the next two steps in the pathway is indole glycerolphosphate synthetase and it is the product of the C locus. The final step in the reaction is the pathway produces tryptophan from indole-glycerol phosphate and serine. This single step is catalyzed by tryptophan synthetase, an enzyme that is a multimer of two proteins that are the product of the B and A genes.
As with all operons, the trp operon consists of the repressor, promoter, operator and the structural genes. In this system, though, unlike the lac operon, the gene for the repressor is not adjacent to the promoter, but rather is located in another part of the E. coli genome. Another difference is that the operator resides entirely within the promoter
Control Circuit for the trp Operon
P/O | L || E | D | C | B | A |
_____________________________________________________
Controlling Region || Structural genes
| trp Operon Gene | Gene Function |
|---|---|
|
P/O |
Promoter; operator sequence is found in the promoter |
|
trp L |
Leader sequence; attenuator (A) sequence is found in the leader |
|
trp E |
Gene for anthranilate synthetase subunit |
|
trp D |
Gene for anthranilate synthetase subunit |
|
trp C |
Gene for glycerolphosphate synthetase |
|
trp B |
Gene for tryptophan synthetase subunit |
|
trp A |
Gene for tryptophan synthetase subunit |
The trp operon is a repressible system. The primary difference between repressible and inducible systems is the result that occurs when the effector molecule binds to the repressor. With inducible systems, the binding of the effector molecule to the repressor greatly reduces the affinity of the repressor for the operator, the repressor is released and transcription proceeds. The lac operon is an example of an inducible system. With repressible systems, the binding of the effector molecule to the repressor greatly increases the affinity of repressor for the operator and the repressor binds and stops transcription. Thus, for the trp operon , the addition of tryptophan (the effector molecule) to the E. coli environment shuts off the system because the repressors binds at the operator.
Inducible system - the effector molecule interacts with the repressor protein such that it can not bind to the operator
Repressible system - the effector molecule interacts with the repressor protein such that it can bind to the operator
Attenuation of the trp Operon
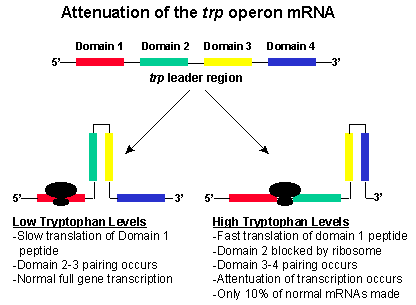
One element of the trp operon is the leader sequence (L) that in immediately 5' of the trpE gene. This sequence about 160 bp is size also controls the expression of the operon through a process called attentuation. This sequence has four domains (1-4). Domain 3 (nucleotides 108-121) of the mRNA can base pair with either domain 2 (nucleotides 74-94) or domain 4 (nucleotides 126-134). If domain 3 pairs with domain 4, a stem and loop structure forms on the mRNA and transcription stops. This structure forms when the level of tryptophan is high in the cell. If domain 3 pairs with domain 2, then the stem and loop structure does not form and transcription continues through the operon, and all of the enzymes required for tryptophan biosynthesis are produce. These events occur when tryptophan is low in the cell.
If domain 4 is deleted, the stem and loop structure can not form and transcription of the remainder of the operon will occur even in the presence of tryptophan. Domain 4 is called the attenuator because its presence is required to reduce (attenuate) mRNA transcription in the presence of high levels of tryptophan.
Domain 1 is also an important component of the attenuation process. The section of the leader sequence encodes a 14 amino acid peptide that has two tryptophan residues.
How does this entire attentuation process work? We will discuss the molecular events that occur under conditions of high and low tryptophan.
trp Operon Transcription Under High Levels of Tryptophan
When the cellular levels of tryptophan are high, the levels of the tryptophan tRNA are also high. Immediately after transcription, the mRNA moves quickly through the ribosome complex and the small peptide is translated. Translation is quick because of the high levels of tryptophan tRNA. Because of the quick translation, domain 2 becomes associated with the ribosome complex. Then domain 3 binds with domain 4, and transcription is attenuated because of the stem and loop formation.
trp Operon Transcription Under Low Levels of Tryptophan
Under low cellular levels of tryptophan, the translation of the short peptide on domain 1 is slow. Because of the slow translation, domain 2 does not become associated with the ribosome. Rather domain 2 associates with domain 3. This structure permits the continued transcription of the operon. Then the trpE-A genes are translated, and the biosynthesis of tryptophan occurs.
Copyright © 1997. Phillip McClean