Non-adiabatic time-domain DFT approach
Dr. Kilina’s group also utilized NA time-domain DFT approach, to study decoherence in nanosystems. Their NA method treats phonons semi-classically, which allows them to explicitly incorporate quantum decoherence effects within the electronic subsystem. It was found, for the first time, that the phonon-bottleneck in CdSe QDs is directly related to the quantum Zeno effect: To achieve the bottleneck, elastic electron-phonon scattering, decohe-rence, must be faster than the coherent electronic transition. These findings are published in Journal of Physical Review Letters (Phys. Rev. Lett. 2013, 110, 180404). In this paper they also confirmed that thin ZnS shell on the CdSe surface provides defects in the interfaces resulted in formation of hole trap states. These traps decouple the electron from the hole, preventing Auger recombination and allowing for fast relaxation. The established relationship between the phonon-bottleneck and the quantum Zeno effect, together with the atomistic details provided by Dr. Kilina’s simulations, generate an understandding of the conditions necessary to achieve and control the phonon-bottleneck experimentally via synthetic manipulations of the QD’s shells, dopants, ligands, packing, etc.
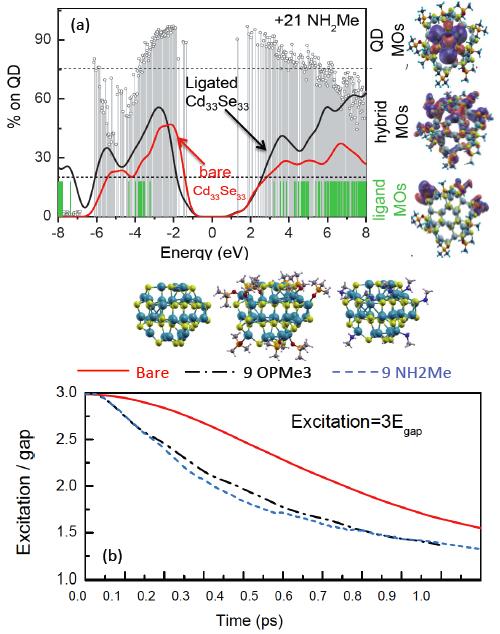
Dr. Kilina’s group also study charge and energy transfer mechanisms in QDs functionalized by metal-organic complexes. New photovoltaic and photocatalysis applications have been recently proposed based on these hybrid systems, while the exact mechanism and direction of the charge transfer controlling their photochemistry is still under debates. DFT-based simulations of the morphology, electronic structure, and optical response of chemically functionalized QDs led Dr. Kilina to predictions of the prerequisite conditions that govern the direction of the charge transfer in these systems. Working in close collaboration with experimentalists from Los Alamos National Lab, they have elucidated the mechanism of attachment of the Ru(II)-complexes to the QD surface (Langmuir 2011, 27, 8377). Dr. Kilina’s computational findings support the conclusion made on the basis of experimental IR spectra of the dye-functionalized CdSe QDs that the complexes attach exclusively to the Cd sites of the QD surface via a bridging geometry, with the carboxylic anchoring group being deprotonated in the process. Her very recent investigations (J. Phys. Chem. C 2013, 117, 18216) revealed that independent of the complex attachment and the type and size of the QD, the complex introduces unoccupied orbitals near the band gap of the QD, while occupied orbitals localized on Ru(II) are deep inside in the QDs valance band. Such an energy alignment of complex and QD orbitals makes the hole transfer from the photoexcited QD to the complex energetically unfavorable, but supports a scenario of electron transfer from the photoexcited complex to the QD. These results were totally obtained by the undergraduate student, Mary Hedrick, who has also significantly contributed in writing the paper. They also found out that change of metal in the complex insignificantly affecting the alignment of the electronic levels of the dye versus the QD’s states, while substitution of a bipyridine to a thiocyanate ligand, results in a shift of the orbital localized on this ligand towards the very edge of the valence band of the QD and make the hole transfer possible. These investigations have resulted in several presentations in national and international conferences (Telluride Conference and ACS Fall 2013) and expected to be submitted to J. Am. Chem. Soc. in the nearest future. As a result of these calculations, Dr. Kilina’s group have determined the prerequisite conditions that govern the direction of the charge transfer in these systems, which is the first step towards future predictions on how to chemically control QD-complex morphology and the QD electronic structure via chemical functionalization in order to improve the charge transport, as well as guide the experimental realization of QD-based solar cells and photo-oxidizer agents.

