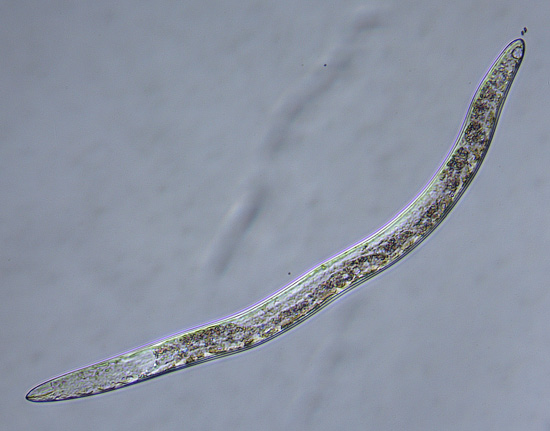
Agrios, G.N. Plant disease caused by nematodes, in Plant Pathology, ed. Agrios, G.N. Elsevier Academic Press, USA. pp 863-864 (2005).
Ayala, A., Allen, M.W., and Noffsinger, E.M. Host range, biology, and factors affecting survival and reproduction of the stubby root nematode. Journal of Agriculture of the University of Puerto Rico. 54, 341-369 (1970).
Franc, G.D., Harveson, R.M., Kerr, E.D., and Jacobsen, B.J. Disease management. in SugarBeet Production Guide, ed. by Wilson, R.G. University of Nebraska, USA. pp 131-160 (2001).
Gray, F.A. Sugarbeet cyst nematode. in Compendium of Beet Diseases and Pests, eds. Harveson, R.M., Hanson, L.E., and Hein, G.L. APS press, USA. pp 70-71 (2009)
Hafez, S. Sampling procedure to diagnose nematode infestations. CIS 1056. University of Idaho, USA. (1997).
Hafez, S. Sugarbeet nematodes in Idaho and eastern Oregon. CIS 1072. University of Idaho, USA. (1998).
Ingham, R.E., Hamm, P.B., Williams, R.E., and Swanson, W.H. Control of Paratrichodorus allius and corky ringspot disease of potato in the Columbia Basin of Oregon. Supplement of the Journal of Nematology. 32, 566-575 (2000)
Mojtahedi, H., and Santo, G.S. Ecology of Paratrichodorus allius and its relationship to the corky ring-spot disease of potato in the Pacific Northwest. American Journal of Potato Research. 76, 273-280 (1999).
Yan, G.P., Khan, M., Huang, D., Lai, X., and Handoo, Z.A. 2016. First report of the stubby root nematode Paratrichodorus allius on sugar beet in Minnesota. Plant Disease 100: 1022.