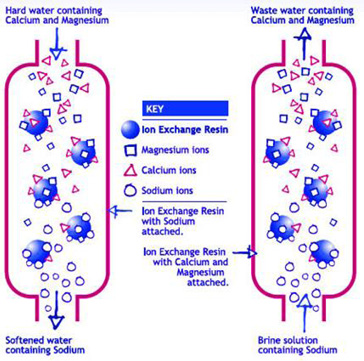
Before buying any water treatment equipment, you should know what impurities are in the water supply. The types and amounts of impurities in your water can be determined by an accredited water testing laboratory. A list of water testing laboratories is available in NDSU WQ1341 “Drinking Water Quality: Testing and Interpreting Your Results.” The results of the water test will help determine if softening is needed. The water test may reveal other dissolved minerals that may require additional treatment.
If you obtain water from a private water supply, water testing is your responsibility. Water testing should be done on a regular basis. If a problem is suspected, test more often.
Community water supplies are monitored and treated to protect users from health threatening water impurities but may still have high hardness. Ask your supplier for a copy of the latest water test results.
Consumption of hard water is actually good for humans since the body will use some of the calcium and magnesium, but for appliances, it is considered a nuisance and not a health problem. Hardness removal is not a necessity to protect your health, but water softening is popular because most people prefer softened water for bathing, cleaning and washing.