Field Guide to Sustainable Production of High-quality Durum Wheat in North Dakota
(A1825, Revised March 2026)Production guide on production and storage of high quality durum wheat.
Andrew Friskop, Extension Plant Pathologist
Brady Goettl, Extension Soil Science Specialist
Clair Keene, Extension Agronomist
Janet Knodel, Extension Entomologist
Rob Proulx, Extension Precision Ag Specialist
Kenneth Hellevang, Extension Ag Engineer - Emeritus
Durum wheat (Triticum turgidum L. var durum Desf.) is one of three classes of wheat commonly grown in North Dakota. Durum wheat acreage in North Dakota varies from year to year based on its market value and price relative to spring wheat and other crops that are adapted to the region.
North Dakota acreage annually averages around one million acres and produces slightly over one-half of US durum. Average yield is approximately 40 bushels per acre.
Durum is a premium market class of wheat used largely for making pasta. The production of pasta requires grain with high protein content, density (hard, vitreous kernels) and gluten strength. Therefore, grain quality is critical in marketing durum. High-quality durum wheat commonly provides a significant price premium, compared with the other market classes of wheat.
Sustainable durum production takes into account the long-term impacts of production practices on the soil and water, and other environmental factors, as well as profitability to the producer. This guide seeks to identify best practices based on information derived from unbiased, scientific research, that are considered to be good for the soil, environment, producer, processor and end-user of the grain.
Field Selection and Crop Rotation
Successful durum wheat production is challenging primarily due to adverse weather and presence of plant diseases. For example, during flowering and early grain fill in humid, wet conditions can favor Fusarium head blight (FHB) because durum does not have a high level of genetic resistance to this disease. Rainfall after maturity and before harvest also is problematic because these conditions can cause the grain to bleach and lose its amber color. For these reasons, most of the durum production in North Dakota occurs in the northwestern quarter of the state, with lesser amounts in the southwest.
If durum is grown in higher rainfall regions of the state, apply the best practices for FHB management and the timing of harvest to avoid the potential for grain quality loss.
Growing durum within an appropriate crop rotation impacts the productivity of the crop and potential for long-term sustainability of the cropping systems. Growing durum after a noncereal crop is highly recommended. The positives for growing durum after a broadleaf crop when compared with another cereal crop include the following:
- Increased yield
- Reduced disease pressure, especially for residue-borne foliar diseases such as tan spot
- Reduced in-field inoculum sources of Fusarium graminearum, the causal organism of FHB, or scab
- Reduced potential for the buildup of soil-borne pathogens
- Ability to use herbicides with different modes of action, which helps delay the buildup of herbicide-resistant/tolerant weeds. This is especially important as limited options are available for some of the crucially important herbicides used for weed control in durum.
When deciding where to place durum in your cropping system, also consider the following:
- When a legume crop proceeds durum, a nitrogen credit of 40 pounds per acre is available and can help reduce fertilizer costs. Following a legume crop also reduces the risk for FHB, compared with following corn or small grain.
- Shallow-rooted and earlier-maturing crops such as peas and lentils generally use less water than other crops and will leave more water for the succeeding crop.
- Deep-rooted crops such as corn and sunflower tend to use more soil water and extract it from deeper depths than other crops. These are not good preceding crops if rainfall is likely to be low or stored moisture is a significant portion of the water used within a cropping season, and when little soil moisture recharge occurred during the winter.
- Corn produces significant amounts of residue. Corn residue harbors Fusarium and increase the problem of FHB in years when the environment is conducive for FHB. Therefore, it is not recommended to grow durum following corn.
- Durum grown for the production of foundation or registered- class seed cannot be planted on land that grew spring, durum or winter wheat during either of the two preceding years.
- Data suggest that in some crop sequences, durum yields may be improved when cereals are grown only once in three years (see data from the Crop Sequence Calculator at
www.ars.usda.gov/ARSUserFiles/30640500/CSC/CSC.htm).
Tillage and Residue Management
Tillage has long been a primary means of managing crop residue, controlling weeds and preparing the seedbed for the crop. Nevertheless, tillage hastens the loss of soil moisture through evaporation and exposes the soil’s surface to greater risk of erosion by wind and water and reduces long-term productivity.
With improved machinery and herbicide technology, no-till planting of durum eliminates the negatives of tillage while still enabling adequate weed control, and excellent seed placement and crop emergence. No-till planting with retained crop residues is one of the most significant management strategies in western cropping regions of North Dakota. It has enabled more intensive cropping, higher yields, better water use efficiency and superior protection of the soil resource.
Additionally, soil health parameters generally improve after a few years of continuous no-till cropping. Water infiltration rates are improved and beneficial biological activity is increased. Nitrogen (N) fertilizer rates are 50 pounds per acre less for long-term no-till production of durum, compared with traditionally cultivated fields, due to enhanced nitrogen efficiency from biological N cycling and reduction of the potential for leaching and denitrification losses.
Retaining residues helps protect the soil from wind and water erosion, growing-season water evaporation from topsoil, and fosters greater microbial activity and diversity within the primary root zone of crops (Figure 1). Previous crop residues have value as a cover and a source for carbon because they are returned to the soil. They are also a valuable source of plant nutrients that will be recycled back into the cropping system through decomposition.
In no-till systems, excessive residue can be problematic, particularly with high-residue crops such as corn. Using choppers and spreaders when harvesting can help produce a more uniform cover and facilitate planting the following season.
Cutting the previous crop high and leaving as much stalk as possible in small grains and corn can help avoid residue matting. Baling and removing straw occasionally (once in three years) in most situations will not negatively impact the sustainability of the system.
Crop rotations that alternate low-residue producing crops with high-residue producing crops are also an effective means of regulating the buildup of residues. Most growers who have been using no-till for a decade or more actually have difficulty with maintaining adequate residue levels due to the tremendous diversity of macro and microorganisms in their system.

Variety Selection
Variety selection is a foundational management component for obtaining high-yielding, high-quality durum wheat. Varietal choice can impact grain yield and quality and the type of management practices that might be needed to optimize profitability. As with all field crops, durum varieties vary in their agronomic and quality characteristics. Moreover, variety performance may interact with the environment, meaning the performance of a variety relative to another variety may change, depending on the environment in which they are grown.
When considering a durum wheat variety, producers should consider the strengths and weaknesses a variety can offer, soil and field characteristics, and weather and disease challenges that might be associated with the crop. The following are some important characteristics to consider:
Yield
When evaluating a variety for its grain yield potential, use data from replicated trials in multiple locations and years. Varieties that do well when the results are averaged from many environments likely will do well in future seasons. These varieties are characterized as stable varieties and are more likely to be the most productive, year after year, when compared with a variety with relatively very high yield in one location per year and only average performance in other environments.
NDSU replicated, multi-location research trial data can be found in the annually produced “North Dakota Durum Wheat Variety Trial Results and Selection Guide” (A1067) available online (https://www.ndsu.edu/agriculture/extension/publications/north-dakota-durum-wheat-variety-trial-results-2025-and-selection-guide) or by using the “Variety Selection Tool” available online (https://vt.ag.ndsu.edu/).
Disease Resistance
Giving the most weight to yield is common when selecting a variety. However, host resistance to diseases can impact the performance of the crop significantly, so consider disease resistance as an important criterion when selecting a variety. Disease resistance ratings for common diseases for most available cultivars can be found in the current selection guide (see the first table in that guide).
Most available varieties have good levels of resistance to leaf and stem rust and moderate resistance to other foliar diseases. Nevertheless, disease resistance genes can break down as pathogens evolve. Even with the ability to manage many fungal diseases with a fungicide, the most sustainable, longer-term strategy is to have the best genetic resistance available in an adapted cultivar.
Moreover, in environments when disease pressure is high, an integrated approach using genetic resistance with a well-timed fungicide results in the best management of a disease. Current levels of genetic resistance to FHB in durum varieties is less than what is available in spring wheat.
The current recommendation is to avoid using any variety rated as very susceptible to FHB. With these varieties, the risk of having severe damage from FHB is high, even if FHB is not that common in your region of the state. If you are growing durum in an environment where FHB has been problematic in the past, use only varieties rated a 5 or less for FHB.
End-use Quality
Durum as a premium market class of wheat has high grain quality standards in the marketplace. Grain protein levels of 13% are the standard for the choice milling grade. Discounts are usual and can be significant when protein levels are below this standard. Conversely, grain protein levels that exceed 14% can generate a substantial premium in some years. Protein is often related to vitreous kernel content (“hard count”). To make choice milling grade, 85% vitreous kernel content is required. The variety selection guide (A1067) includes seed protein and vitreous kernel data among durum varieties.
The environment and the variety grown impact grain protein content at harvest. Although the environment is hard to predict, reaching high levels of protein is more difficult when conditions are favorable for high yield; the nitrogen to produce the grain exceeds the amount of nitrogen provided, so the protein content is reduced.
Varieties vary in their ability to produce protein in a given environment. If you routinely have difficulty in reaching 13% in your environment, and the protein premium or discount appears that it will be high at harvest and for higher yielding environments, consider selecting a variety that is known to have high protein levels.
In NDSU field studies evaluating the influence of variety, planting date and seeding rate of new durum wheat varieties, characteristics such as protein content, falling number, kernel yellow pigment content and gluten index were more dependent on the particular variety than the environment in which it was grown.
Bleaching of the bright amber color of durum can occur due to weathering, which also can result in discounts at the elevator. No published data are available on varietal differences for weathering.
Low falling numbers (below 315 to 330) are another source of discount. Low falling numbers are an indication of sprout damage in a seed lot and generally are a problem when rainfall or very high humidity levels occur after the grain has matured and before harvest. Delayed harvest has a negative effect on yield and other quality parameters due to prolonged exposure to the environment.
Some varieties resist preharvest sprouting better than others. Those with the highest falling numbers are those that are the most likely to resist preharvest sprouting; however, rainfall occurring when the crop is mature can still cause sprouting in a field grown to a variety with good falling number.
Cadmium Accumulation Potential
Cadmium (Cd) is a heavy metal that can be toxic to humans. Durum wheat is one of the few cereals that accumulates Cd when grown in soils with high levels. Varieties are now available that have reduced accumulation of Cd. Varieties with this trait should be grown if your soils are high in Cd and if exceeding the market threshold for Cd has been problematic. These varieties are identified in the variety selection guide (A1067).
Certified Seed
Certified seed is seed that has been inspected and tested by the State Seed Commission, which then is able to certify that the seed is a known variety, the seed lot is genetically pure, free from prohibited weed and other crop seeds, and free of certain seed-borne diseases. Further, the seed is certified as to its germination percentage at the time that it was certified.
With certified wheat seed, you purchase the purest genetics, meaning the seed is assured to germinate well and be free from noxious weeds and harmful diseases. If you keep your own seed for future plantings, you should purchase certified seed at least every three years to help maintain the genetic purity of the variety grown.
Also be sure to conduct a germination test every year to know the germination of the seed lot you will plant. Seed lots with less than 80% germination may show a loss of vigor in the field. Additionally, be wary of seed lots with low falling numbers (below 300) or high DON levels, or that were frosted during grain filling because these factors are known to negatively impact germination and vigor. For a list of certified seed dealers, consult the latest “North Dakota Field Seed Directory” available online (https://www.seed.nd.gov/field-seed-directory).
Seeding Date and Rate
Seeding date and rate of durum wheat can impact grain yield and quality and associated economic return. Temperature, humidity and rainfall at key growth stages of durum greatly impact subsequent grain quality.
One key growth stage of durum is flowering. During this time period, if environmental conditions are favorable, severe FHB levels can occur. All durum varieties are susceptible to FHB, and staggering seeding dates is one option of reducing the risk that all fields will be damaged by this disease when environmental conditions are favorable to its development during a specific period of the growing season.
Also, high temperatures at flowering can negatively affect fertilization. By optimizing the seeding date (early season), producers may be able to reduce the risk associated with flowering during July, when higher temperatures are expected.
Durum typically is seeded later than many of the other early season crops. However, a reduction in yield may be experienced. NDSU seeding date trials found that the earliest date at which producers were able to plant resulted in a significant yield increase over later dates (Figure 2). Subsequent, two-week delays in seeding from the first date continued to significantly decrease yield.
In addition to yield, test weight also declined as the seeding date was delayed.
Early seeding dates also will enable producers to harvest in August, when the threat of rainfall and damage to mature grain is lower.
Seeding rates of durum are similar to those of other small-grain crops. Lower seeding rates may result in the production of more tillers, while higher seeding rates tend to produce fewer tillers and increase the chances of lodging.
NDSU research indicates that a plant population resulting from seeding 1.2 million pure live seeds (pls) per acre resulted in the highest average yield, compared with seeding 900,000 and 1.5 million pls per acre. Also, data suggest that a significant cultivar-by-seeding-rate interaction may result for yield in environments that are below 52 bushels per acre. In lower yielding environments, an increase in the seeding rate above 1.2 million seeds tended to increase grain yield. Overall, the seeding rates evaluated had limited effect on the agronomic and quality traits for the cultivars evaluated.
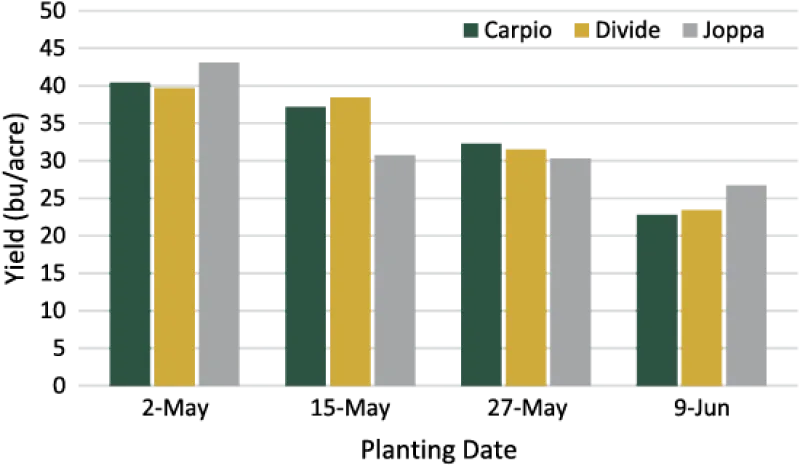
Seed Treatment
Seed treatments are an important tool in durum wheat production and help manage pests that affect stand establishment. Fungicide seed treatments are most effective when managing seed-borne diseases such as loose smut and common bunt.
Fungicide seed treatments can also be used to manage early season Fusarium crown rot, common root rot and Pythium root rot. Seed-applied fungicides should not be viewed as providing season-long protection because the residual of a fungicide lasts only a couple of weeks after planting.
Insecticide seed treatments are effective in managing soil-dwelling insects such as wireworm. Most seed treatment packages will be broad spectrum, combining multiple fungicides and potentially an insecticide.
The decision to use a seed treatment should include several factors and may not always be needed. Refer to the “North Dakota Field Crop Plant Disease Management Guide”
(https://www.ndsu.edu/agriculture/extension/publications/north-dakota-field-crop-plant-disease-management-guide) and
“North Dakota Crop Insect Management Guide”
(https://www.ndsu.edu/agriculture/extension/publications/north-dakota-field-crop-insect-management-guide) for more information on labeling and efficacy.
The use of seed treatments does not guarantee a higher plant stand. A positive stand establishment response with the use of a seed treatment is more likely to be observed in the following situations: seed lots harboring a seed-borne pathogen; early planting into cool, wet or otherwise undesirable soil conditions); planting durum following another small-grain; or planting into a field with a known history of root rots or wireworms. Similarly, yield responses are not always consistent because durum can tiller to help compensate for stand loss.
FHB causes losses in quality and yield and also increases the presence of scabby seed. Although not ideal, if scabby seed is to be used as a seed source, it should be cleaned and conditioned thoroughly to remove the majority of the scabby kernels. Also, a germination test should be conducted to check the viability of the seed and a fungicide seed treatment is strongly suggested.
Another common head disease resulting in a contaminated seed source is ergot. Fungicide seed treatments do not manage ergot, and cultural practices such as crop rotation and incorporating ergot sclerotia below the soil surface can help lower the risk.
Fertility Management
Providing adequate levels of plant-essential nutrients is a component of sustainable durum production. Soil testing and NDSU published recommendations are the best guide.
Applying adequate nitrogen (N) is extremely important. Also, phosphate seed-placed or near seed-placed starter is crucial to highest durum yield. Phosphorus (P) and potassium (K) recommendation charts are based on soil test results. Other nutrients are required only if specified on the soil test report. Refer to the NDSU publication “Fertilizing Hard Red Spring Wheat and Durum” (https://www.ndsu.edu/agriculture/extension/publications/fertilizing-hard-red-spring-wheat-and-durum) for current recommendations. This publication covers recommendations for the nutrients likely to be limiting North Dakota durum production.
Additionally, to determine the most economical rate of nitrogen to apply, use the North Dakota Crop Nitrogen Recommendation Calculator (https://lamp.ndsu.edu/soils/FourCropsNitrogenCalculatorWebApp/). It takes into account the location of your farm, soil organic matter content and residual nitrogen, nitrogen credits for the previous crop, and the price of nitrogen and wheat. Soil testing is also a key input into this calculator. The nitrogen calculator is also available as a downloadable app for iPhones and Android phones. Search for North Dakota Crop Nitrogen Calculator for the free download.
Overusing nutrients is expensive and can cause runoff from the farm. Phosphate and N movement into surface water resources are particularly harmful and can result in damage of the water body through algal blooms. The death and subsequent decomposition of those blooms result in eutrophication, or depletion of dissolved oxygen. Some algal blooms also release toxins that are harmful if ingested to livestock, people and wildlife.
The 4Rs (right rate, right time, right source, right place) of fertility management help guide the sustainable use of fertilizers and include applying the right amount of the required nutrient at the right time, using the right source in the right place.
N timing and placement
N is not only the most commonly used fertilizer; it is also the most likely to be lost from the soil before the crop can use it and, therefore, requires special consideration. The general rule of thumb to minimize N losses is to apply most of the N closest to the time of greatest use.
Applying N fertilizer prior to late durum vegetative stages, when N uptake accelerates, is challenging. Therefore, apply most or all of the N needed for the crop prior to planting.
Spring N fertilizer applications are preferable to fall applications. However, fall applications are acceptable on loam soils or in areas not prone to spring flooding after snowmelt.
Anhydrous ammonia and urea are the two N sources that can be fall-applied in the state. Anhydrous ammonia application should not begin before Oct. 1, and then only after soil temperatures measured at the 4-inch depth fall to 50 degrees F between 8 and 10 a.m.
Subsurface banded urea can be applied a week after this date. Broadcast and incorporated urea should wait two weeks after the anhydrous ammonia date. Surface application of urea to no-till acres is not as efficient as subsurface application. The risk of volatility of ammonia is high if at least ½ inch of rain does not fall for several days after application.
Risk is greatest on a soil surface with abundant residue, soil pH greater than 7, high humidity or a light rain. No-till wheat growers should apply urea beneath the soil surface for greatest efficiency, possibility in a one-pass operation at planting.
Liquid N sources, such as 28% N liquid fertilizer (commonly called UAN, or “28”), also should be applied below the surface. If spring conditions prevent below-surface application, banding the 28% on the surface may delay volatilization several days, compared with broadcast application.
For broadcast urea application made to no-till, consider using a urease inhibitor with an effective rate of NBPT (such as Agrotain™ or Limus™) to help reduce volatilization losses. Use streamer bars or similar nozzles to apply UAN in-season prior to jointing if additional N is needed. Application at later stages are less effective for increasing yield, although the timing may help enhance protein levels.
Foliar N for Enhancement of Grain Protein and Vitreous Kernels
A foliar application of N immediately after flowering (post-anthesis) can be used to enhance the grain protein content and vitreous kernel percentage (hard count) of durum. North Dakota research has shown that the best timing of this management strategy is just after flowering is complete on the majority of main stem heads.
The recommended rate is 30 pounds of N per acre as UAN (10 gallons per acre of 28%) mixed with 10 gallons per acre of water. This application should be made in the cool of the day. Some leaf burning will result, but if the application is made immediately post-anthesis, yield reduction is unlikely based on North Dakota field studies.
Addition of herbicides, fungicides or insecticides with the N fertilizer may increase the intensity of leaf burn and limit the efficacy of the pesticide and fertilizer. Do not apply N with fungicides at the flag leaf stage or anthesis because the burning at this time may reduce yield. The use of low rates of slow-release liquid N products before or after anthesis has not been shown to increase grain protein.
Generally, a 0.5% to 1% protein increase has been achieved using post-anthesis N application. In a NDSU durum field study with soil N not sufficient for achieving the milling durum threshold level for protein or vitreous kernel content, protein improved to 13.6% and hard count improved to 96% with post-anthesis N compared to the N check at 11.9% and 84%, respectively (https://www.ndsu.edu/agriculture/ag-hub/publications/carrington-research-extension-center-2024-annual-report).
Potassium
Response of durum to K is unlikely when soil test levels are greater than 100 parts per million (ppm). However, due to the chloride (Cl) contained in potash (0-0-60, potassium chloride) crop yield response to this fertilizer may be realized in fields with Cl levels less than 40 lb per acre.
Historically, most soils in North Dakota had high enough K levels to support excellent wheat production; however, more K-intensive crop rotations (corn and soybean) have led to falling K soil test levels across the state. Current K fertilizer recommendations are based on a soil test critical level of 100 ppm. The recommendation in higher-testing soils is provided to replace K that the crop will remove and to provide chloride if necessary. If Cl values are adequate and other crops in the rotation regularly receive K fertilizer, then potash application when K and Cl are in the high range category are not necessary.
Sulfur
Sulfur (S) is an important secondary nutrient. The S soil test is not a good predictor of possible S deficiency. A more useful method to determine whether soils within a field likely will be S deficient (Figure 3) is to consider which soils are sandy loam or coarser textured, with less than 3% organic matter on higher landscape positions. If rainfall is high before seeding, even durum grown on higher organic matter soils and soils with more clay can be susceptible to S deficiency.
If soils with these properties are present, review fall and spring rainfall conditions. If normal to greater than normal rainfall or greater than normal winter snow was received on these soils in the fall or early spring, the stage is set for possible S deficiency and a pre-emptive spring fertilizer application is recommended and should include a soluble S fertilizer. Ammonium sulfate at rates of about 10 pounds per acre or gypsum at 20 pounds per acre are excellent sources of S.
Elemental S, even premium bentonite-blended forms, will not be nearly as useful in correcting a deficiency as the more soluble forms. Composite-blended granules of phosphate fertilizers that include S could be used, but rates need to be high enough to supply the 10 pounds per acre needed as the ammonium sulfate portion of the fertilizer, or the application should be supplemented with a sulfate containing fertilizer.

Weed Management
Weeds are pests that are present in every field and every season regardless of previous management. Weeds are controlled through chemical, cultural and physical methods.
A broad array of herbicides is registered for use in durum.
See the current “North Dakota Weed Control Guide”
(https://www.ndsu.edu/agriculture/extension/publications/north-dakota-weed-control-guide) for details of products and their recommended rates and timing.
Herbicide-resistant weeds have become a major threat to the sustainability of herbicides as a weed control option. Delaying the evolution of herbicide-resistant or tolerant weeds to existing herbicides is of paramount importance.
Strategies to Minimize Herbicide-resistant Weeds
Weed resistance cannot be prevented, but it can be delayed, and no single strategy is likely to be totally effective. Herbicide rotations, tillage and herbicide tank mixtures with different sites of action can delay resistance by the length of time the selection pressure for a given herbicide is replaced by an alternative control method.
Resistance may occur first in fields where a single site-of-action herbicide is used repeatedly in a growing season or across several growing seasons. Integrated weed management uses multiple strategies to manage weed populations, including:
- Diversify crop sequences with different life cycles. For example, winter annual crops (winter wheat), perennial crops (alfalfa) and summer annual crops (spring wheat, corn or beans), which result in different planting and harvest times, enable more herbicide options and allow varied cultural practices to decrease the risk of the development of herbicide-resistant weeds. Consider weed biology and ecology. Use tillage, crop sequence, soil fertility, planting date, crop competition, weed seed longevity, cover crops and response to herbicides to increase success in weed management.
- Scout fields before and soon after herbicide application. Correctly identify weeds. Use effective herbicides, handweeding, cultivation or tillage, and other methods of weed control to kill weeds that escape or germinate after chemical application. Scout fields at the end of the season and draw field maps to denote locations of weed species, weed density and weed escapes. Save maps as a field record.
- Consider weed biology and ecology. Use tillage, crop sequence, soil fertility, planting date, crop competition, weed seed longevity and response to herbicides to increase successful weed management.
- “Don’t forget the PRE”. Apply effective PRE herbicides at full rates and include multiple mechanisms of action. PRE herbicides will reduce weed emergence and allow flexibility in POST herbicide timing. Residual PRE herbicides applied to soil and early POST (if labeled) will suppress weed emergence through canopy closure, particularly those with a long germination pattern (waterhemp). Use PRE herbicides that will effectively control problem weeds.
- Apply effective POST herbicides. Apply herbicides that include multiple mechanisms of action in tank-mix or in sequential applications. Two or more herbicides in mixture must have activity against potentially resistant weeds to be effective. Herbicides in most commercial mixtures do not target the same weed species. Effective tank-mixtures on weeds will reduce selection of herbicide-resistant biotypes more successfully than rotating herbicide modes of action. Antagonism may occur with some mixtures, especially between contact and systemic herbicides.
- Use high herbicide rates and effective adjuvants. Full rates kill weeds with low-level resistance and dead plants cannot produce resistant progeny. Reduced rates allow plants with low-level resistance to survive, hybridize, and produce progeny with elevated resistance. Hybrid plants (>1 resistance gene) express a higher level of resistance and require even higher herbicide rates to kill the plant. Dead weeds means zero tolerance (no seed production, zero resistant progeny) and is effective resistance weed management.
- Spray small annual weeds. Generally, small weeds (less than 3 inches) are more susceptible to herbicides than large weeds. Even weeds with low level herbicide resistance are more susceptible at 1 inch than at larger growth stages.
- Practice Zero Tolerance. Scout fields after row closure and kill uncontrolled weeds. Seed from escaped weeds will contribute to the weed seedbank and will require diversified weed management strategies of mowing, cultivation or tillage, and hand weeding to achieve near 100% weed control. Timely cultivation can improve weed control and handpulling is effective for single plants or small patches.
- Control weeds in field perimeters, drown out, and non-crop areas. Weeds surviving a partial herbicide dose on field borders can be a repository for the introduction of resistant weeds into a field. Control weeds in all areas of the field where the crop is not growing including field edges, fence lines, waterways, ditch banks, and areas where the crop has either not been planted or has been destroyed.
- Rotate herbicides with different mechanisms of action in consecutive years. Diverse crop rotations can introduce herbicides with different mechanisms of action to delay herbicide resistance. A mix of dead plants, unaffected plants, and plants showing intermediate responses indicate herbicide resistance has occurred.
- Clean tillage and harvest equipment to ensure weed seed will not be transported between fields. This is particularly important in crops that are harvested with a platform header equipped combine.
- Evaluate weed management at the end of each season and revise to improve weed control the next year.
Disease Management
Several diseases have the potential to reduce yield or quality of durum wheat. In many cases, genetic, chemical and cultural methods are available to reduce the impacts of these diseases. Integrating multiple methods results in the best management of a disease.
The most common diseases impacting durum are stripe rust, tan spot, Stagonospora nodorum blotch, bacterial leaf streak and Fusarium head blight.
Rust
Leaf and stem rust are important diseases of durum, but most durum varieties in North Dakota have high levels of resistance. Leaf and stem rust pathogens have the ability to evolve quickly, and new races could overcome resistance in North Dakota varieties.
Many of the current durum varieties are susceptible to stripe rust (yellow rust). Stripe rust is favored by cooler temperatures and prolonged moisture periods and sporadically occurs in the major durum production regions.
The spores of wheat rust pathogens are carried by southerly winds into the state. Therefore, the earlier a rust disease is reported in North Dakota, the greater the chance of yield loss.
Fungal Leaf Spot Complex
The fungal leaf spot complex is associated with the pathogens that cause tan spot, Stagonospora nodorum blotch, and Septoria tritici blotch (Figures 4 and 5). These pathogens overwinter on small-grain residue and spores are released as conditions are favorable in the spring and summer (Figure 6). Spores are dispersed by wind and infect durum in a wide range of temperatures.
During wet growing seasons and prolonged moisture periods, such as dew, large numbers of conidia form in the disease spots and can be carried by wind and rain splash to other leaves to form new infections. Prolonged wet periods of 24 hours or longer can result in spore germination and infection on leaves of durum.
Crop rotations significantly reduce the initial inoculum of fungal leaf spot pathogens because initial spores released from residue do not travel great distances. Rotation of durum with any broadleaf crop will reduce the risk of infection by these three leaf spot fungi. Tillage to bury infested residue or straw management at harvest to aid residue decomposition can be helpful in reducing inoculum levels but is not always an option in durum production areas.
Fungicides are available for early-season and late-season management of fungal leaf spots. NDSU research has shown modest yield responses with the application of a fungicide to wheat for control of early season tan spot when wheat was planted into wheat residue, a susceptible to moderately susceptible variety was grown and when spring rains or dew favor disease development. Early season application is not recommended in the absence of disease or in an environment unfavorable to the development of disease.
Several fungicides are labeled for management of fungal leaf spots. Products containing active ingredients belonging to the FRAC 3, 7 and 11 have very good to excellent management of fungal leaf spots. Consult the most current edition of PP622, “North Dakota Field Crop Plant Disease Management Guide,” https://www.ndsu.edu/agriculture/ag-hub/ag-topics/crop-production for an updated list of registered fungicides and their use rates.



Bacterial Leaf Streak
Bacterial leaf streak (BLS) is caused by the bacterium Xanthomonas translucens pv. undulosa and has been observed frequently in recent years in North Dakota. Yield losses due to BLS have been estimated to range from zero to up to 6%, depending on the stage of infection and susceptibility of the variety. The causal bacterium is primarily seed-borne and can survive on crop debris. The bacterium is spread by splashing or wind-driven rain and enters the plant through wounds or the stomata.
Symptoms of this disease are most noticeable in areas that have had frequent storms associated with high winds and leaf damage. Early leaf symptoms of bacterial streak are characterized by translucent, water-soaked streaks. As the lesions mature, water-soaked streaks become dry and turn brown or necrotic. Additionally, the causal bacterium may be visible as a shiny glaze (clumps of dried bacteria) on the leaf surface within the chlorotic and necrotic lesions (Figure 7).
The primary means of managing BLS is through the use of genetic resistance. The pathogen is seed-borne, so the use of clean seed can prevent infections and spread. However, because of the widespread prevalence of this disease in North Dakota in residue and soil, prevention of BLS through clean seed may have only limited impact. No effective chemical seed treatment or foliar plant protection product is available to manage BLS.
Most durum varieties currently grown in North Dakota are susceptible to BLS but some varietal differences have been observed. See the Durum Selection Guide for the most recent ratings, if available.

Fusarium Head Blight
FHB is the disease that has had the most impact on the durum industry in North Dakota. Since the 1990s, the reduction of durum production in the northeastern part of the state can be attributed to an increase in the frequency and intensity of FHB.
FHB is a fungal disease that can occur on all small grains but is most commonly seen on spring wheat, winter wheat, durum and barley. Although FHB can cause severe yield losses, the major impact of this disease on durum is on its quality.
Yield losses occur from floret sterility and infection results in shriveled, light test-weight kernels. Quality reductions occur when the fungal pathogen produces mycotoxins (deoxynivalenol, or DON) in infected seed.
The primary symptom of FHB is premature bleaching of the wheat head, or spike, that appears 14 to 21 days after flowering. The partly white and partly green heads are diagnostic for the disease in durum (Figures 8 and 9). Additional indications of Fusarium infection are pink to salmon- orange spore masses of the fungus often seen on the infected spikelets and glumes during prolonged wet weather.
Many infected durum kernels are shriveled, lightweight and dull grayish or pinkish. These kernels sometimes are called “tombstones” because of their chalky, lifeless appearance (Figure 10). If infection occurs late in kernel development, Fusarium-infected kernels may be normal in size but have a dull appearance or a pink discoloration and still have DON. Infected kernels of durum often lose their amber translucence and appear chalky or opaque.
FHB is best managed by integrating multiple management tools. The use of a single tool often fails when the environment favors severe disease. Management strategies to reduce FHB should include a combination of as many of the following practices as possible:
Genetic Resistance. None of the commercially available durum varieties have a high level of FHB resistance. Nevertheless, resistance differences occur among varieties, and variety selection has a significant impact on FHB severity. As mentioned in the section on variety selection, avoid varieties that are rated 7 to 9 in the selection guide and consider using only the most resistant varieties in areas prone to FHB development such as areas of higher moisture and humidity. around flowering). Using varietal resistance and a well-timed fungicide results in the best management of FHB.
Tillage and Crop Rotation. Tillage practices that bury residue from small grains or corn can reduce the inoculum potential of the fungus. In minimum or no-till practices, effective spreading and distribution of chaff and other residue may allow faster decomposition of the chaff, reducing inoculum potential. Chopping or grinding corn residue to reduce the size of the remaining stalk pieces favors more rapid disintegration of host tissue. Crop rotation is effective in reducing in-field FHB levels. Planting durum on last year’s broadleaf ground can reduce inoculum levels significantly. The greatest risk of Fusarium infection occurs when small grains are planted on last year’s small-grain or corn residue. Although tillage and crop rotation help reduce the inoculum level, they will not prevent the development of the disease if environmental conditions are favorable. Fusarium spores can be carried by wind from other fields and from previous residues that have not yet decomposed to initiate an infection.
Planting Date. Stagger the planting of durum fields and use varieties differing in days to maturity; this reduces the risk of a producer’s entire crop flowering or going through early grain fill during a period favorable for Fusarium infection.
Fungicide. A fungicide can reduce FHB damage, particularly if combined with genetic resistance. Reductions in FHB severity of 50% to 60% can be achieved when fungicides are applied at early flowering. The most effective active ingredients for FHB and DON suppression are prothioconazole, metconazole and pydiflumetofen. See NDSU Extension publication PP622, “North Dakota Field Crop Plant Disease Management Guide for an updated list of registered fungicides and their use rates.
Disease Forecasting. The NDSU small-grain disease forecasting system provides information for the risk of fungal leaf spot, rusts and FHB at the following website: www.ag.ndsu. nodak.edu/cropdisease/. A multi-state FHB forecasting model for FHB also is available (www.wheatscab.psu.edu); it will provide FHB risk for the following six days.
Both forecasting systems are useful tools to indicate the possible need for a fungicide application. The forecasting systems are developed using weather parameters including humidity, temperature, precipitation and leaf wetness, to estimate risk.
The grower must decide if the variety grown is susceptible to the leaf disease or FHB, if good yield potential is present to warrant fungicide use and if the weather site chosen is representative of the weather in the vicinity of the wheat field.
Integrated Management. Durum growers will manage several diseases during a growing season, and using an integrated management strategy is the best approach. Several of the aforementioned diseases can be managed by multiple tools, and employing several of them will reduce the development of diseases. Avoiding the reliance on one management tool will reduce the ability of a pathogen to adapt through host and fungicide resistance and prolong the use of the most effective management tools.



Insect Pest Management
Several insects can cause damage to durum crops. Aphids, armyworms, cutworms, grasshoppers and wireworms are examples of insects that are problematic to a wide range of crops. Information about these insects and treatment guidelines is found in the current “North Dakota Field Crop Insect Management Guide,” https://www.ndsu.edu/agriculture/extension/publications/north-dakota-field-crop-insect-management-guide. Wheat stem sawfly and wheat midge are the two insects that can cause economic damage to durum and other classes of wheat in North Dakota.
Wheat Stem Sawfly – Cephus cinctus Norton (Hymenoptera: Cephidae)
The wheat stem sawfly is considered one of the most significant insect pests of wheat. Sawfly populations fluctuate across years and locations, with the most significant damage occurring in the western half of North Dakota.
Sawfly populations generally have increased since 2005. Recent surveys suggest yield losses due to this pest are in the range of 10 to 25 percent statewide, but losses of 100 percent have been reported in a heavily infested field.
Wheat stem sawfly is a wasp-like insect (Figure 11). The adult is about ¾ inch long and is relatively short-lived (seven to 10 days). Adult emergence occurs over a long period, typically about three weeks, but sometimes as long as one month. The first emergence typically occurs in mid-to-late June in North Dakota.
Female sawflies deposit eggs into the elongating stems of host plants in early summer, and developing larvae feed on stem tissue and move up and down the length of the stem. (Figure 12) Although several eggs may be laid within a stem, only a single larva survives to maturity due to the cannibalistic behavior of the larva.
As the plant matures, and usually before harvest, the larva moves down to the base of the stem and chews a notch around the inside of the stem. The notch weakens the stem, which usually breaks. Much of the sawfly damage is associated with lodging and harvest losses. Still, significant losses in kernel numbers and size can also occur due to larvae feeding on the vascular tissues of the stem during grain filling (Figure 13).
Spring wheat, winter wheat and durum wheat are the main cereal crops attacked by wheat stem sawfly, although infestations in other small grains, such as barley, triticale and spelt, have been observed. Barley suffers minimal damage, and cultivated oats do not support wheat stem sawfly development, although females do lay eggs in oats.
Several cultural control practices used singly or in combination may help reduce wheat stem sawfly populations. Swathing, tillage, delayed planting, and crop rotation have all been recommended, although each has an associated cost.
Swathing and using a stripper header are the only pest management practices that can be utilized in the current year of the infestation. Swathing sometimes is conducted on just the outer one or two swaths bordering the field when the infestation is heavy only in the field edges. Swathing prevents sawfly larvae from cutting the stems and reduces yield loss due to lodging.
The disadvantages of this technique are that it requires an additional field operation, and swathing may adversely impact parasitic wasps that attack sawfly larvae in the upper portions of the stems. If you decide to swath a durum crop, use a high swathing height (leave at least the lower third of the plant stem) to conserve the parasitic wasps that attack wheat stem sawfly.
To determine if you should swath a field, calculate the percent of plants infested by sawflies before harvest. The presence of wheat stem sawfly can be verified by splitting stems and looking for the S-shaped larvae inside the wheat stem, or by observing the sawdust-like frass within the wheat stem. Infested wheat stems often have a darkened area on the stem just below the nodes as a result of the internal feeding from the sawfly.
If sawflies infest more than 15% of stems, the field should be swathed. Swathing sawfly-infested wheat is recommended as soon as kernel moisture drops below 40% to prevent infested stems from lodging.
Stripper headers may be used for straight cutting the crop. This header will pick most wheat stems off the ground. Stems that are not firmly attached will be brought into the combine, while stems still firmly attached to the ground will have grain stripped from the stem.
Fall and spring tillage have been used to expose overwintering sawfly larvae to cold and dry conditions, thereby increasing larval mortality. Nevertheless, tillage for sawfly control runs counter to current recommendations for reduced or no-tillage, and it usually does not increase mortality sufficiently to reduce the sawfly infestation level the following year.
Delayed planting (after May 20) can be partially effective if it delays the crop sufficiently so it has not reached the stem elongation stage when female sawflies are ready to lay eggs. This practice may result in reduced yield due to the adverse effects of late planting on grain filling.
Rotation with a nonhost crop can reduce populations within a field. However, sawflies are weak flyers and usually fly short distances. Re-infestation from nearby fields, grass borders or Conservation Reserve Program land is possible in subsequent years. Crop rotation can also decrease the negative impacts of diseases, as well as increase the benefits of soil fertility. Crop rotation to a non-host is highly recommended for fields where sawfly damage was observed the previous year.
Breeding solid-stem spring and winter wheat cultivars has imparted good levels of sawfly resistance. Unfortunately, no solid-stemmed durum varieties are available.
Insecticides have generally been ineffective against the wheat stem sawfly. The egg, larval, and pupal stages are well-protected inside the plant stem, and the adult emerges over a long period, reducing the chance of exposure to a single insecticide application.
Biological control of wheat stem sawfly is common, and parasitism levels can be as high as 88%, with an average of 35%. In North Dakota, the most important species in wheat fields is Bracon cephi, a parasitic wasp. These parasitic wasps attack and kill larvae of wheat stem sawfly in the stem. Any use of insecticides will have a detrimental impact on parasitic wasps.



Wheat Midge – Sitopdiplosis mosellana (Géhin) (Diptera: Cecidomyiidae)
Wheat midge is a significant pest of North Dakota spring wheat and durum wheat, causing significant yield losses and lower grain quality. Wheat midge can be found anywhere wheat is grown in North Dakota, especially in the northern half of the state. Recently, wheat midge has become a more occasional insect pest in sporadic areas.
The adult midge is a small, orange, mosquito-like fly that is active from late June to late July (Figure 14). Peak emergence is usually near mid-July. Wheat is susceptible to egg laying by female wheat midge from head emergence through mid-flowering. The orange larvae hatch from the eggs and feed within the wheat florets, reducing kernel weight and grain quality (Figure 15). Mature larvae drop to the soil to overwinter as cocoons in late summer. There is one generation per year.
Pest management strategies for wheat midge include wheat midge emergence degree-day modeling, field scouting, insect trapping, the use of insecticides, cultural control, biological control, the development of midge-tolerant varieties and risk forecasting for the wheat midge population the following year.
Degree Day Modeling. The wheat midge degree-day model predicts adult wheat midge emergence, enabling producers to determine when to scout fields and assess the risk of infestation to their wheat crop based on their planting date. Producers can access the wheat midge degree-day model on the North Dakota Agricultural Weather Network (NDAWN) website at
https://ndawn.ndsu.nodak.edu/wheat-growing-degree-days.html.
Based on North Dakota field observations, midge larval infestations are the highest when wheat heading occurs during peak female emergence at 1,475 degree-day (DD) accumulations. When using the wheat midge base temperature, 40 degrees F, for wheat development, heading occurs around 1,000 to 1,100 DD accumulations (typically monitored at 32 degrees F).
The adult wheat midge emergence can be forecasted based on the degree-day accumulations after wheat planting. At 1,300 DD accumulation 10% of wheat midge females have emerged, at 1,475 DD accumulation, at 50% of wheat midge females have emerged, and at 1,600 DD accumulation 90% of wheat midge females have emerged.
Select your nearest NDAWN station and enter your spring wheat planting date. The output indicates the expected growth stage of the wheat, whether the growth stage is susceptible to midge infestation, and the timing and percentage of wheat midge emergence.
Field scouting. To determine the level of infestation, durum heads should be scouted from head emergence through mid-flowering by walking a W-pattern in the field and checking five sites, examining 10 heads per site. Monitor fields at dusk (9 p.m. and later) when temperatures are above 60 degrees and wind speed is less than 6 mph. By using a flashlight, the orange wheat midge can be counted as they flutter around the wheat heads.
Insect trapping. Pheromone traps and sticky traps are also used to monitor adult wheat midge in wheat fields (Figure 16). These traps can alert producers to the presence of wheat midge and provide an indication of relative population density; however, trap captures do not indicate whether an insecticide treatment is warranted. Pheromone traps and insect lures can be purchased from commercial insect trapping suppliers, such as Great Lakes IPM (www.greatlakesipm.com).
Insecticide management. Insecticide is only effective in controlling the adult wheat midge. Insecticide control for larvae, which feed on the developing kernels, has not been demonstrated. Larvae are protected inside the glumes during foliar insecticide applications.
Economic threshold. Insecticide treatment is warranted when scouts or producers observe one or more wheat midge adults for every seven or eight durum heads.
For registered insecticides for wheat midge control, refer to the current “North Dakota Field Crop Insect Management Guide,” www.ndsu.edu/agriculture/extension/publications/north-dakota-field-crop-insect-management-guide.
Cultural Control. Crop rotation away from continuous wheat will help reduce local populations of wheat midge. Non-susceptible crops to wheat midge include oats, barley, and broad-leaved crops such as canola, dry edible beans, pulse crops, soybeans, and sunflowers. Crop rotation to a non-host is highly recommended for fields where wheat midge damage was yield-reducing the previous year. Early planting will also help the wheat plant complete flowering before the peak emergence of wheat midge.
Biological Control. Parasitic wasps provide natural biological control of wheat midge larvae. One of the most common parasitic wasps in North Dakota is Macroglenes penetrans. Past survey work indicates that the average parasitism rate of wheat midge is 22%, with a range of 0% to 100% in fields.
Populations of parasitoids fluctuate and tend to follow the populations of wheat midge by a one-year lag period. NDSU Extension Entomology recommends avoiding late insecticide applications after mid-flowering to minimize the negative impacts on parasitic wasps that are active at that time.
Resistant Wheat Varieties. The resistance gene Sm1 is the first gene identified as conferring resistance to the wheat midge through antibiosis. The Sm1 gene is available in durum wheat varieties in Western Canada, such as ‘AAC Succeed,’ ‘AAC Marchwell,’ and ‘CDC Carbide.’ Future breeding efforts should focus on placing the Sm1 gene into wheat varieties developed in North Dakota.
Risk Forecasting. An annual wheat midge risk map is available to producers based on the NDSU Extension soil survey conducted by county Extension agents and specialists in 21 counties in northern North Dakota with a history of infestations. Soil samples are examined for overwintering wheat midge larval cocoons and used to forecast infestation risk for the following year. Parasitism rates by wasps are also assessed to evaluate natural biological control.
Wheat midge risk and parasitism maps are available at: www.ndsu.edu/agriculture/ag-hub/wheat-midge-risk-maps.
Because soil surveys over the past 10 years have detected few wheat midge cocoons. NDSU Extension Entomolgy will shift to pheromone traps for monitoring wheat midge and its parasitoid wasp. These traps are more sensitive for detecting low population and help determine emergence timing to guide scouting in areas with higher populations.



Drying and Storage
After you have invested money and effort in producing grain, make sure you protect that investment by drying and managing grain properly.
Clean Bins Before Harvest
Grain and fines, small pieces of broken grain, weed seeds and chaff, remaining from previous crops almost always contain stored grain insects. To reduce insect problems in the new crop, avoid mixing old and new grain. Also, clean bin walls and floors, and the area outside of bins to remove old grain dust, fines and kernels. The space under perforated floors typically contains material conducive for insects, but it is very difficult to clean. Fumigate the underfloor area if an insect infestation occurred previously.
At least two weeks before placing grain in the bin, consider using a bin spray by applying a coarse spray of an approved insecticide to the point of run-off. Pay close attention to areas that might hide insects, such as under false floors and in vents. Cracks around doors and vents may serve as sources of infestation.
Manage Fines
Fines and foreign material such as weed seeds cause problems in grain storage because they are more susceptible to mold and insect attack than are whole kernels, they restrict airflow during aeration, and they tend to concentrate under fill sprouts during grain handling. Fines management begins at harvest. Select and use grain harvesting and handling equipment to minimize the amount of fines in stored grain and consider using a grain cleaner to remove fines before grain is stored.
Research has shown that in addition to improving grain storability, cleaning scab-infected grain over a gravity table reduces deoxynivalenol (DON) levels in the cleaned grain.
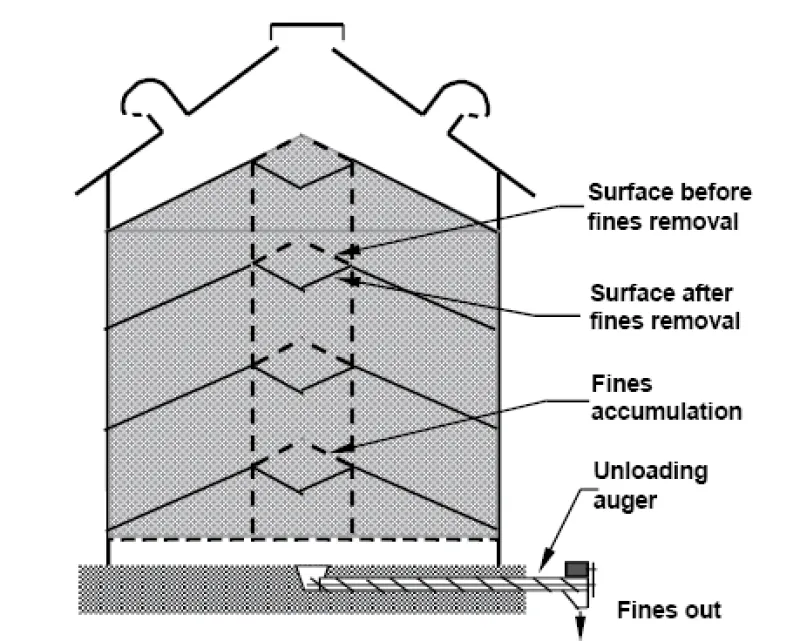
Fill bins in a way to limit concentration of fines at the center. Either use a grain spreader to attempt to distribute fines throughout the bin, or periodically withdraw fines from the center as the bin is filled (Figure 17). An alternative is to remove some grain after the bin has been filled, which is commonly referred to as “coring” the bin. However, because the grain unloading occurs in a funnel shape, the width of fines removed from the center is narrow.
Control Moisture
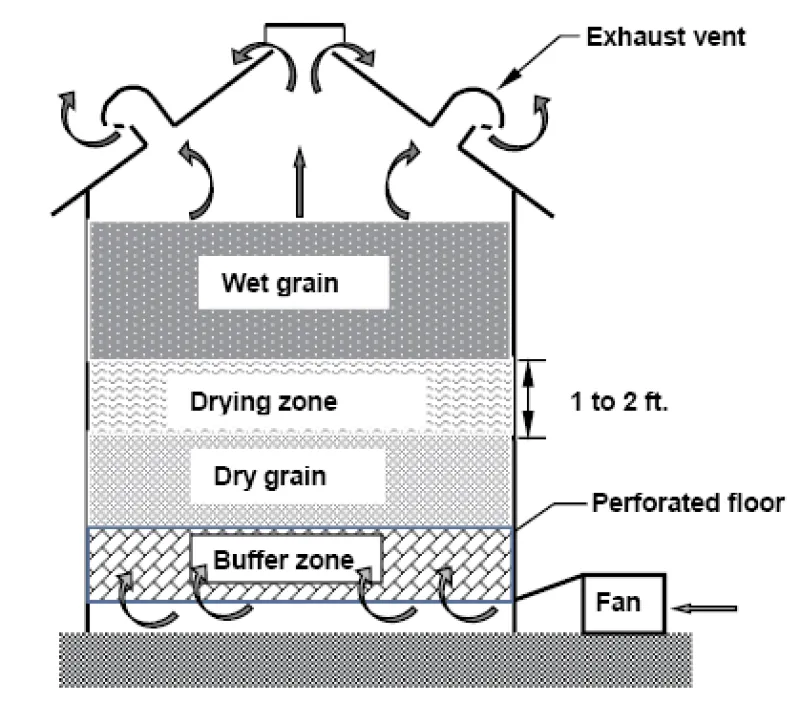
Molds and insects need moisture to live and reproduce, so make sure grain is dry before it is stored. The maximum recommended storage moisture content for durum wheat is 13.5% (wet basis). If grain is too wet when it is harvested, you can dry it with natural, unheated air if your bin is equipped with a perforated floor and a large enough drying fan.
The recommended airflow rates for natural-air drying durum wheat are 0.5 cubic feet of air per minute per bushel of grain (cfm/bu) for up to 16% moisture grain, 0.75 cfm/bu for up to 17% moisture grain and 1 cfm/bu for up to 18% moisture grain (Table 1).
Drying times will be longer with cooler temperatures. Drying time is related to the airflow rate, so increase the airflow rate to reduce the drying time. A typical airflow rate is 0.75 cfm/bu, which permits drying up to 17% moisture durum in a reasonable time. In general, operate natural-air drying fans 24 hours per day until all grain in the bin is dry unless fog is occurring, rain is falling, or the average daily temperatures drop below about 40 degrees F.
No drying will occur if the equilibrium moisture content (EMC) of the air, calculated as the average of the daily maximum and minimum temperatures, exceeds the grain moisture. So do not operate the fans if the EMC continues to exceed the grain moisture. The fan heats the air 3 to 5 degrees, which reduces the relative humidity about 3% to 10%. The increase in temperature and reduction in humidity cause grain to dry an additional 0.75 to 1 percentage point.
Adding supplemental heat to increase the air temperature about 5 degrees will permit you to continue drying the grain during the fall while outdoor temperatures average above 40 degrees. Table 2 lists the grain moisture contents (EMC) that can be attained based on the ambient temperature and relative humidity when using natural-air drying.
Table 1. The recommended airflow for natural-air and low-temperature durum wheat drying and drying times, assuming air at 70 degrees F and 60% relative humidity.
| Moisture content (% wet basis) | Minimum Airflow (cfm/bu) | Drying Time (days) |
| 16 | 0.50 | 42 |
| 17 | 0.75 | 31 |
| 18 | 1.00 | 25 |
If drying is not completed in the fall, run fans as needed to keep grain at 20 to 30 degrees during the winter, and resume drying during the spring when the outdoor air temperature averages above 40 degrees, which is typically during May. Figure 18 illustrates the natural-air drying process.
Table 2. Equilibrium moisture content (% wet basis) for durum wheat exposed to air at various temperatures and humidities. Grain moisture content will be a little lower due to the fan heat.
| Temperature (°F) | Relative humidity (%) | |||
| 20 | 40 | 60 | 80 | |
| 40 | 7.9 | 11.1 | 14.0 | 17.3 |
| 50 | 7.7 | 10.8 | 13.6 | 16.9 |
| 60 | 7.5 | 10.5 | 13.2 | 16.5 |
| 70 | 7.3 | 10.2 | 12.9 | 16.1 |
| 80 | 7.1 | 10.0 | 12.6 | 15.7 |
Durum wheat also can be dried in high-temperature dryers designed for use with corn and other grains, but you need to reduce the drying air temperature to prevent grain damage. To limit the potential effects of high temperatures on the chemical structure of the grain, keep grain temperature below 140 degrees when drying grain used for seed.
The typical maximum drying air temperature for drying milling durum is 150 degrees for 16% grain moisture and 130 degrees for 20% grain moisture. The allowable temperature will vary, depending on dryer design.
Aerate to Control Grain Temperature
Dry grain should be cooled to less than 60 degrees as soon as possible after harvest by operating aeration fans during cool weather. In late summer, this might mean running fans only at night.
Do not worry too much about high nighttime relative humidity during aeration cooling as the relative humidity of the air is reduced as the air cools the grain. In addition, grain temperature changes much faster than does moisture content, so little grain is affected in the amount of time required to cool the grain.
Cool the durum whenever the average outdoor temperature is 10 to 15 degrees cooler than the grain temperature. The goal is to cool the grain to below 50 degrees as soon as possible.
In late fall or early winter, use aeration fans to cool grain to 20 to 30 degrees for winter storage. If grain is not stored at less than 20 degrees during winter, you should not need to run fans to warm the grain to 30 to 40 degrees in the spring. Operate fans in late winter and spring to remove solar heat gain and natural warming of the grain. The goal during the spring is to keep the grain at 30 to 40 degrees.
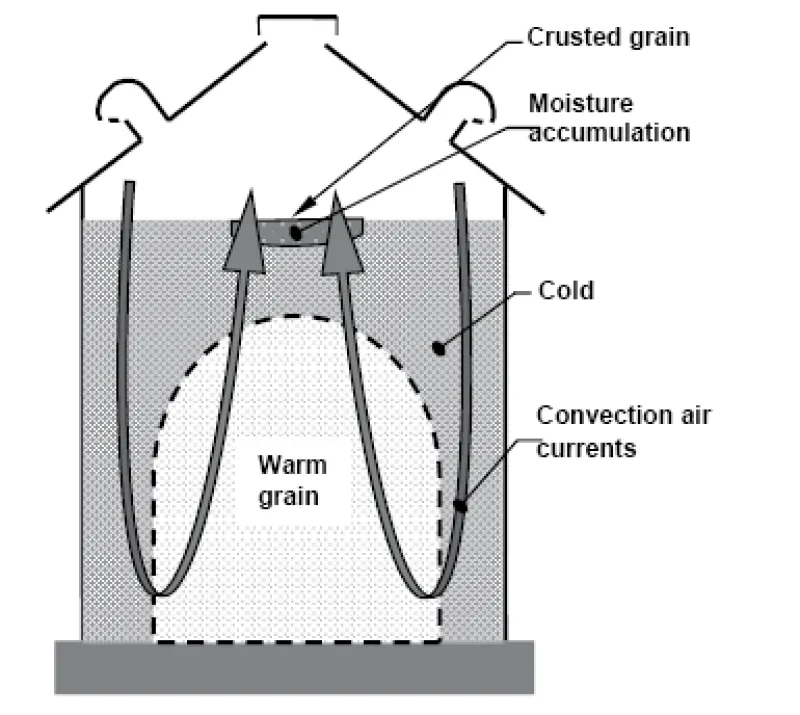
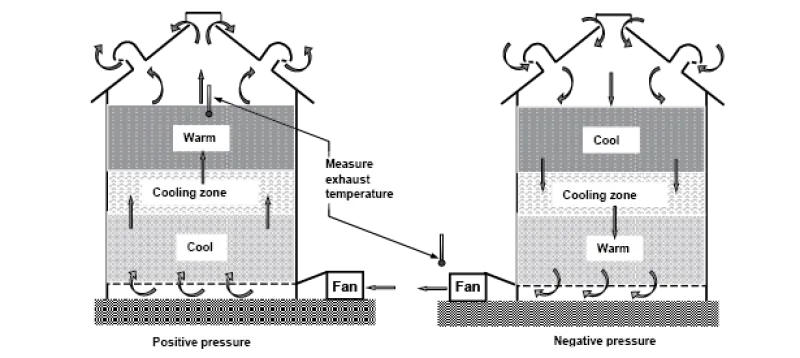
Cooling grain limits mold and insect activity, and it reduces moisture migration. Moisture migration can result in rewetting and eventual spoilage of grain at the top center of inadequately cooled bins (Figure 19).
You can estimate the number of hours a fan must be operated to cool a bin of grain by dividing 15 by the airflow in cfm/bu. For example, in a storage bin that has an airflow of 0.2 cfm/ bu (a typical value for farm bins), cooling the grain takes about 75 hours (15 ÷ 0.2), or approximately three days, of fan operation. Figure 20 illustrates how grain cooling occurs and how to check for completion of cooling.
Check Stored Grain Regularly
Check stored grain at least every two weeks when experiencing warm weather and until the grain has been cooled for winter storage. Measure grain temperature and moisture, look for moldy, discolored or crusted kernels, and check for signs of insects. During the winter when grain temperature is near 30 degrees, checking the grain at least every three to four weeks should be adequate.
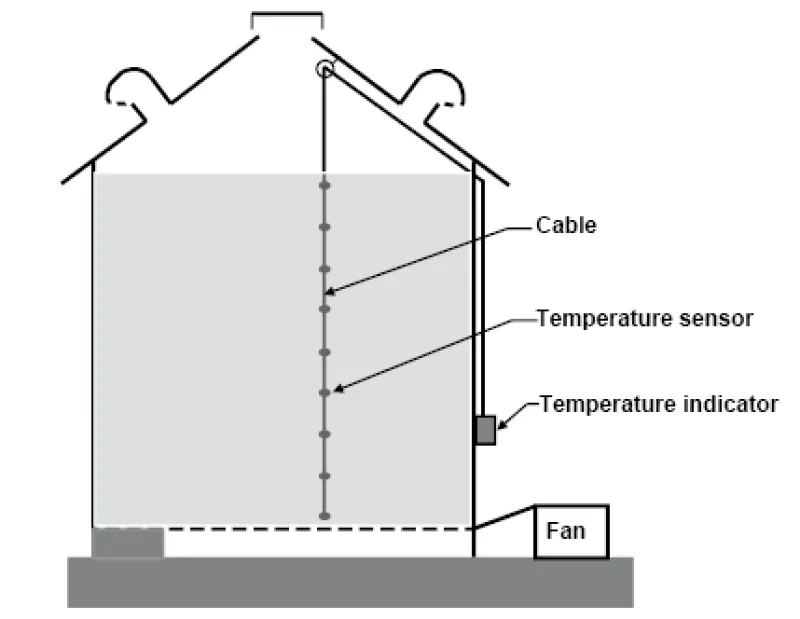
Temperature cables can make checking grain temperature much easier, but they are not a substitute for examining the grain (Figure 21). The grain temperature can differ significantly just a few feet away from the cable.
When checking stored grain, start the fan briefly and smell the first air to leave the bin for musty or sour odors. If you detect problems, run aeration fans to cool the grain. If aeration does not control the problem, unload the bin and clean, dry, feed or sell the grain.
Wear a mask designed to filter mold spores, especially if you know the grain is moldy, to avoid health problems caused by mold spores. Also, grain dust is a health hazard. The mask normally will have a rating of N-95 or N-99.
Storage of Scab-infected Grain
University of Minnesota research on the storability of grain infected by FHB indicated that infected grain deteriorated slightly faster in storage than did grain that had been cleaned on a gravity table to remove scab-infected kernels. The difference in storability between cleaned and uncleaned grain was relatively small.
The fungal species that causes scab infection and produces DON remained viable during storage at 18% and 20% moisture, but that fungal species died during storage at 16% moisture. This means that DON production during storage is unlikely for wheat stored at less than 16% moisture.
This publication was authored by Joel Ransom, former Extension Agronomist; Greg Endres, Extension Cropping Systems Specialist - Emeritus; Shana Forster, Director, NDSU North Central Research Extension Center; Andrew Friskop, Plant Pathologist; David Franzen, former Extension Soil Science Specialist; Richard Zollinger, former Extension Weed Specialist; Kirk Howatt, Associate Professor, NDSU Plant Sciences; Janet Knodel, Extension Entomologist; Patrick Beauzay, Research Specialist; and Kenneth Hellevang, Extension Agricultural Engineer - Emeritus, 2017.
Durum wheat (Triticum turgidum L. var durum Desf.) is one of three classes of wheat commonly grown in North Dakota. Durum wheat acreage in North Dakota varies from year to year based on its market value and price relative to spring wheat and other crops that are adapted to the region.
North Dakota acreage annually averages around one million acres and produces slightly over one-half of US durum. Average yield is approximately 40 bushels per acre.
Durum is a premium market class of wheat used largely for making pasta. The production of pasta requires grain with high protein content, density (hard, vitreous kernels) and gluten strength. Therefore, grain quality is critical in marketing durum. High-quality durum wheat commonly provides a significant price premium, compared with the other market classes of wheat.
Sustainable durum production takes into account the long-term impacts of production practices on the soil and water, and other environmental factors, as well as profitability to the producer. This guide seeks to identify best practices based on information derived from unbiased, scientific research, that are considered to be good for the soil, environment, producer, processor and end-user of the grain.
